Accelerator Technology
Unlocking therapeutic cell migration
MicroCures’ cell movement accelerator program targets Fidgetin-Like 2 (FL2), a key microtubule regulatory protein (MRP) that naturally decreases the movement of cells.
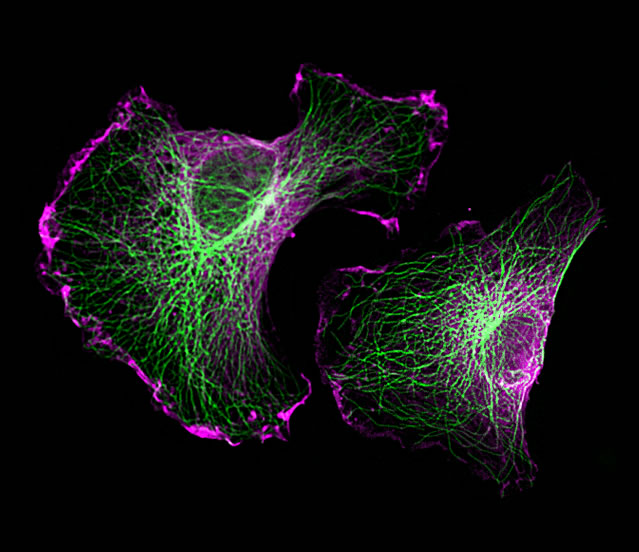
Microtubules (green) and FL2 (purple) in migrating human cells. FL2 naturally suppresses cell movement by severing microtubules.
Under normal circumstances, FL2 selectively severs microtubules at the cell’s leading edge which suppresses the rate and directionality of cell movement. However, when inhibited, FL2 causes cells to shift into a accelerated movement state.
Due to its significant impact on cell movement, MicroCures believes that the ability to silence FL2 activity has the potential to play an important therapeutic role in enhancing and accelerating tissue, nerve and organ repair. Based on this, the company is currently developing topical/local siRNA-based inhibitors of FL2 to promote the regeneration/repair of the cornea, skin, and cavernous nerve.
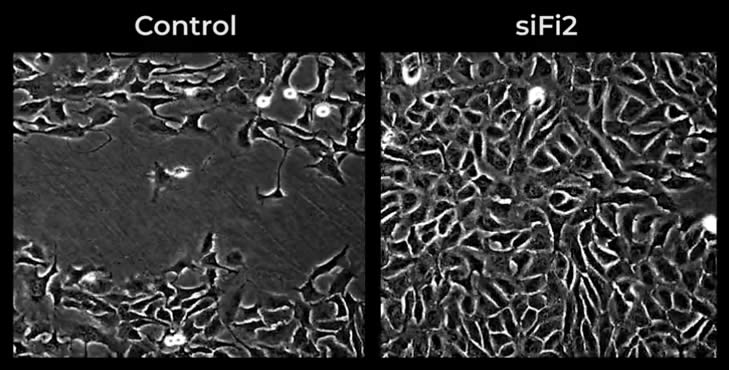
When FL2 is inhibited, cells migrate with enhanced speed and direction as compared to normal FL2 expression.
“Depletion of FL2 from mammalian tissue culture cells results in a more than two-fold increase in the rate of cell movement, in part due to a significant increase in directional motility.”
Conclusion from in vitro study conducted by researchers, including Dr. David Sharp, MicroCures’ founder and CSO
Transformative platfom for repair and regeneration
MicroCure’s proprietary cell movement accelerator platform is a fundamentally new and disruptive technology. It provides a much-needed alternative to today’s complex, potentially unsafe and often unproven regenerative medicine treatments that are used for wound healing and nerve/tissue regeneration. Unlike regenerative medicine approaches that rely on engineered materials or systemic growth factor/stem cell therapeutics, MicroCures’ technology simply directs and accelerates the body’s natural healing processes through local, temporary modulation of cell movement.

